| Project Acronym | EATRIS-Plus |
|---|---|
| Funding Programme | Horizon 2020 |
| Budget | 4,900,000 EUR |
| Coordinator | EATRIS |
EATRIS-Plus Project ran 1 January 2020 – 31 December 2023 (48 months). The project has now concluded and the information on this website is being updated to highlight the opportunities that our community can continue engaging in. While we are working on these updates, we encourage everyone to explore the Multi-Omics Toolbox (MOTBX), The Patient Engagement Resource Centre (PERC) and the Innovation Management Toolbox (IMT). Also don’t forget to take a look in the Publications and Deliverables section below for journal articles, pre-prints and other deliverables of the project.
In case of questions about specific opportunities contact the Project Manager via the contact form at the end of the page.
Ongoing Initiatives
News and Highlights
EATRIS-Plus in a nutshell
Our flagship project EATRIS-Plus aims to build further capabilities and deliver innovative scientific tools to support the long-term sustainability strategy of EATRIS as one of Europe’s key research infrastructures for Personalised Medicine.
The main goals of EATRIS-Plus are to:
- Consolidate EATRIS capacities in the field of Personalised Medicine (particularly omics technologies) to better serve academia and industry and augment the number of EATRIS Innovation Hubs with large pharma;
- Drive patient empowerment through active involvement in the infrastructure’s operations;
- Expand strategic partnerships with research infrastructures and other relevant stakeholders, and
- Further strengthen the long-term sustainability of the EATRIS financial model.
Publications and Deliverables
Artificial Intelligence Enabling Improved Patient Treatment Strategies – Challenges and Recommendations
Preprint | Zenodo | December 2023
Quality criteria for global expansion of the repository of multi-omic reference values
Deliverable | Zenodo | December 2023
Multi-omics Quality Assessment in Personalized Medicine through EATRIS
Preprint BioRxiv | November 2023
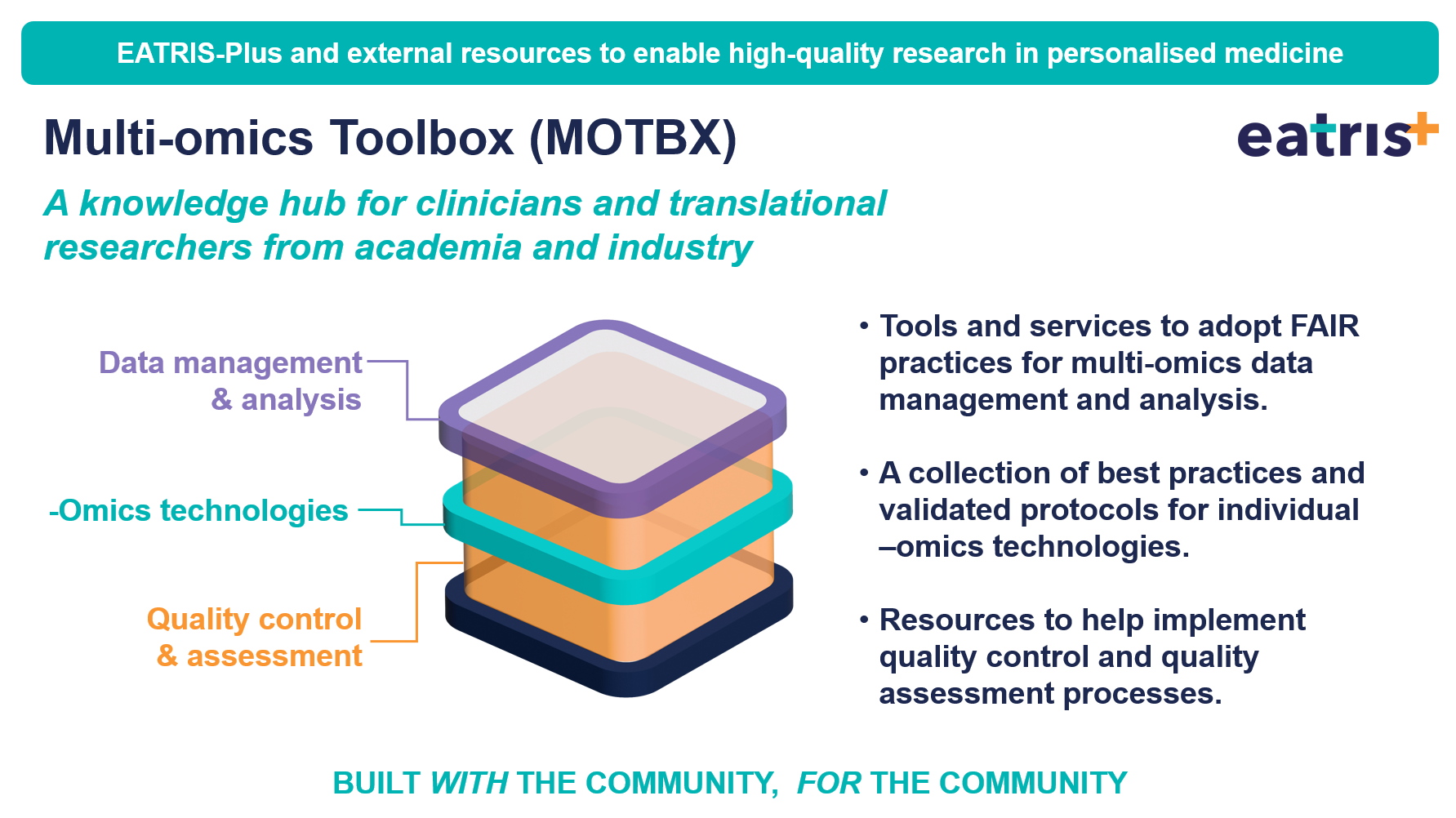
The Multi-omics Toolbox (MOTBX): Empowering Multi-omics Research and Analysis for the Translational Medicine Community
Deliverable | Zenodo | November 2023
Metabolomic predictors of phenotypic traits can replace and complement measured clinical variables in population-scale expression profiling studies
BMC Genomics | July 2022
Computational approaches for network-based integrative multi-omics analysis
Frontiers Molecular Biosciences | November 2022
FAIR Genomes metadata schema promoting Next Generation Sequencing data reuse in Dutch healthcare and research
Nature Scientific Data | April 2022
Tackling the translational challenges of multi-omics research in the realm of European personalised medicine: A workshop report.
Frontiers Molecular Bioscience | October 2022
Evaluation of Methods for Improving Comparability of Cross-Omics Data from Different Cohorts
Deliverable
Technological Reference Protocols for Transcriptomic, Proteomic and Metabolomic Analysis
Deliverable
Past Events
Sharepoint Login
Members of the EATRIS-Plus Project can enter the SharePoint platform here