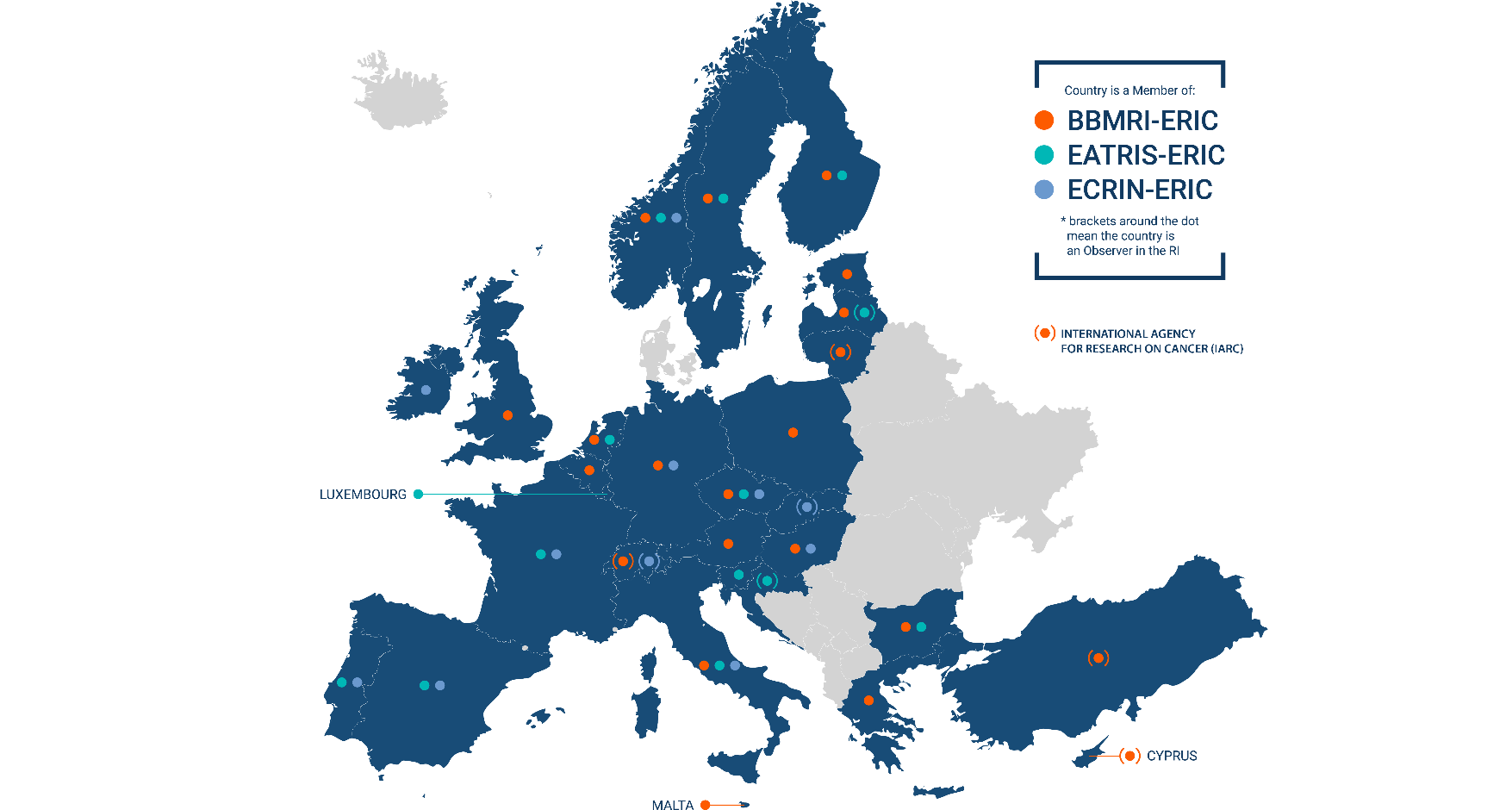
BBMRI, EATRIS and ECRIN, the medical European research Infrastructures, supported by the European Strategy Forum on Research Infrastructures (ESFRI), have joined forces by forming the European Alliance of Medical Research Infrastructures (EU-AMRI). EU-AMRI aims to facilitate the effective and efficient use of scientific services, expertise and tools by academia and industry for the seamless translation of their scientific discoveries into new treatments and solutions for patients.
The three infrastructures share a commitment to increasing patient benefit of research, promoting good research practice and ensuring greater access to available infrastructure resources. The end goal of EU-AMRI is to make it easier for the broader research community to achieve their goals by delivering scientific services with the highest standards of quality and reproducibility.
Via a new website www.eu-amri.org, EU-AMRI allows industry and academia to conveniently choose from a range of services according to their needs. These services span the biomedical pipeline, from preclinical models to sample access, biomarker research to multinational clinical trials and product development. In addition, the research infrastructures offer extensive training and support in ethical, legal and societal issues (ELSI), quality management, regulatory affairs, public-private collaborations and more.
In March 2022, EU-AMRI will be formally launched online and in Brussels (see event page here). The programme will include a presentation of EU-AMRI and will be followed by a round-table discussion with important stakeholders for the future of the EU-AMRI Alliance.
For more information and to subscribe to the launch event, see here. Pre-registration is open here.
About EU-AMRI
The European Alliance of Medical Research Infrastructures (EU-AMRI) is the result of the collaboration between three European research infrastructures, namely EATRIS-ERIC (focused on translational medicine), ECRIN-ERIC (focused on multinational clinical research) and BBMRI-ERIC (focused on biobanking and biomolecular resources). It aims to provide complementary services to researchers in the field of biomedical sciences and support the development of personalised medicine and new treatments. Established in 2020, EU-AMRI comprises over 700 research institutions and university hospitals, 29 European Countries as Members or Observers, working to improve the research process and tackle unmet medical needs. EU-AMRI continues to play a meaningful role in the global response to COVID-19, dealing with challenges related to diagnostic tests, biomarker development, development of treatments and vaccines for COVID-19.