| Website | www.istitutotumori.na.it |
|---|---|
| Address | Via M. Semmola 80131 Napoli Italy |
| Platforms | ATMP, Biomarkers, Imaging & Tracing, Small Molecules |

Brief description of the Centre and its history
The Istituto Nazionale Tumori IRCCS “Fondazione G. Pascale” (INT – Pascale) is the largest Clinical Care and Research Cancer Center in Southern Italy. The mission is the prevention, diagnosis and care of cancer and its combination with innovative research in oncology. The organizational model is based on that of US “Comprehensive Cancer Centres” in which multidisciplinary teams are dedicated in an integrated manner to tackle all aspects related to the care of cancer patients.
INT – Pascale was founded in year 1933 and in 1940 received the first recognition as research center. It is composed of four distinct buildings: 1) Main Hospital and Surgery (186 beds); 2) Day Hospital and Surgery (42 day hospital beds); 3) Research Building; 4) Administrative Building. Furthermore INT – Pascale owns an outstation entirely dedicated to Research and located in Mercogliano (AV). INT – Pascale has approximately 800 employees of which 203 medical doctors, 50 biologists and 338 nurses.
Brief description of the main research activities
The research activities of the INT – Pascale are organized in macro-areas. This organization has the purpose of focusing the activities of INT – Pascale on major issues, to promote interactions between researchers from diverse fields, and to create a “critical mass” of projects that involve different expertises. The Macro-areas where research activities of the INT-Pascale are currently organized are called:
- Prevention and Risk Factors in Neoplastic Disease
- Tumor Markers and Innovative Diagnostic Procedures
- Integrated Treatments in Oncology
- Innovative Therapeutic Strategies in Advanced Disease
The scientific productivity over the past two years has increased significantly and is now constituted by more than 280 publications/year in journals with an impact factor totaling approximately 1,200 points. Research activities are being financed both by funds made available by the Ministry of Health and by National and International competitive grants from various institutions. In the last years, several International patent applications have been filed by researchers of the INT – Pascale which cover newly discovered diagnostics or therapeutics and recently the Institute has fostered the creation of two spin-off SMEs.
A characterizing theme of INT – Pascale is represented by the strong commitment towards clinical experimental studies. INT – Pascale has an independent Ethical Committee which has approved more than 588 clinical studies (including both experimental and observational) between years 2007-2015. In 2015 about 1100 patients have been enrolled in experimental trials.
Finally, in the last years the INT – Pascale has strongly invested in the set of a strong pharmacogenomic platform for molecular diagnostic and has become a reference center in Italy and Europe for Quality assessment of several molecular biomarkers for response to therapy (KRAS, EGFR, BRAF).

Core Facilities (clinical or research)
- Pathology Lab with associated Biobank
- Centralized Clinical Pathology Laboratory
- Centralized Radiology
- Centralized Radiotherapy
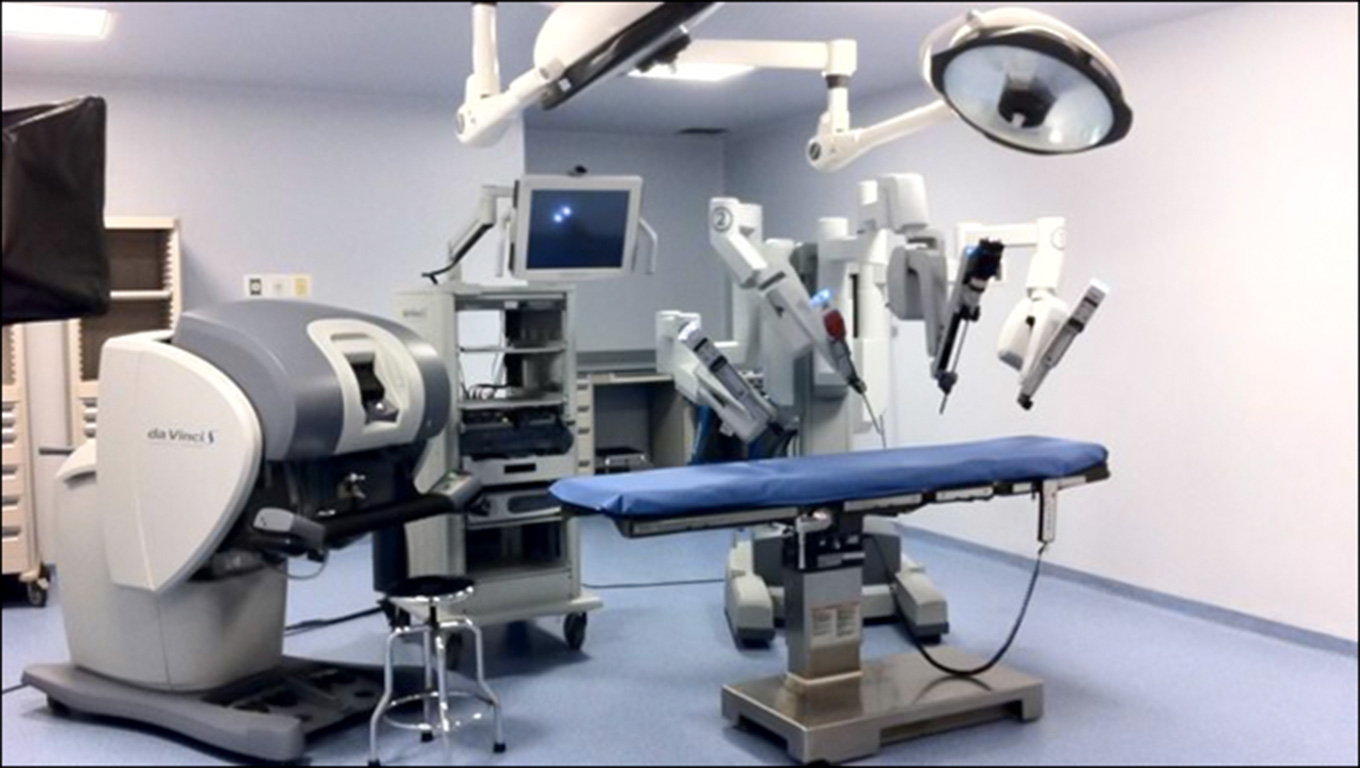
- Minimally invasive surgery (Robotized “da Vinci” Surgical System)
- Pharmacogenomic facility
- Proteomics/metabolomics facility
- Animal facility
- Cyclotron and Radiopharmacy
- Phase I facility
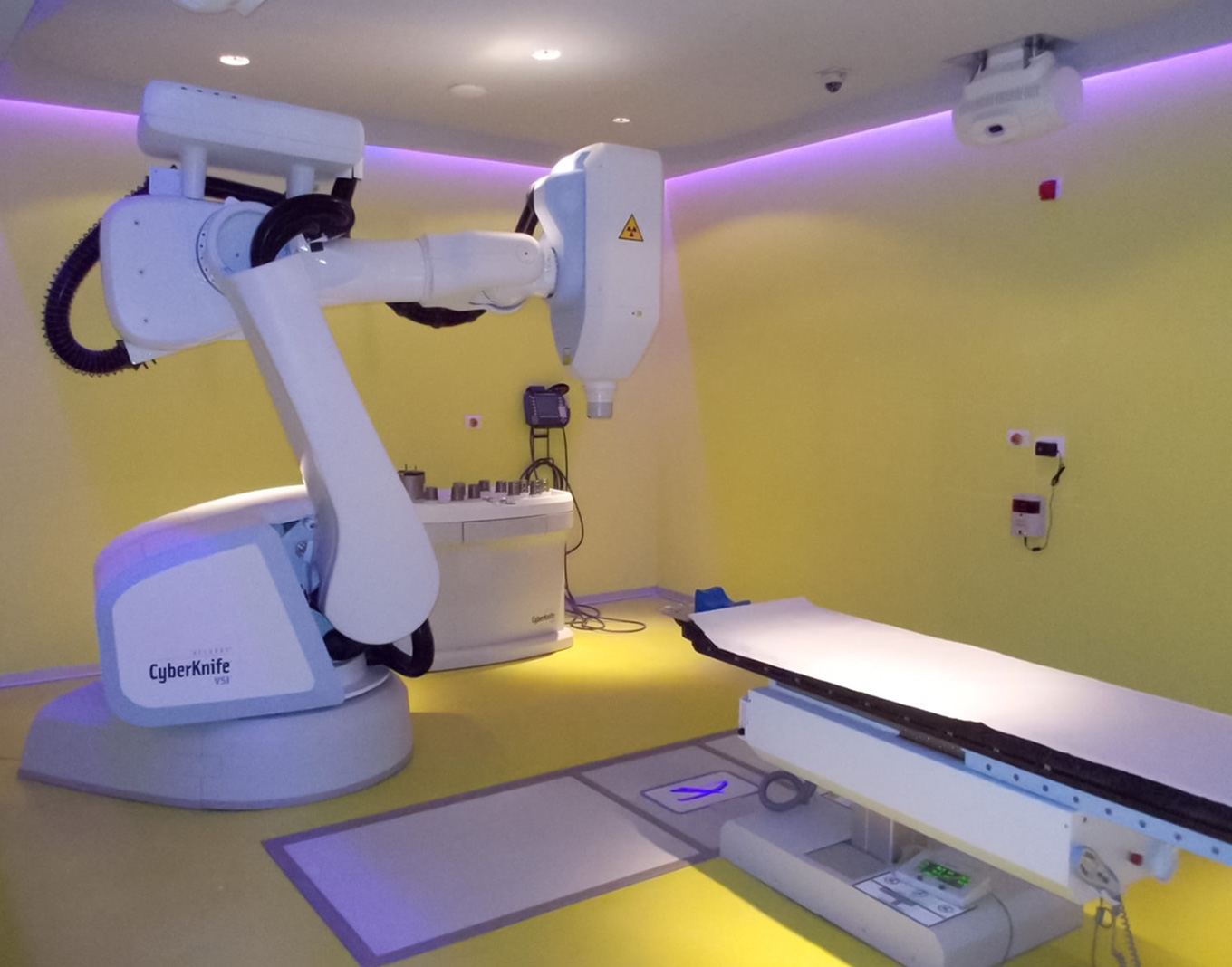
INT-Pascale is investing in continuous technological improvements. In recent years, thanks also to the contributions in capital account from the Ministry of Health, the technological level of the equipment has improved thanks to the purchasing of: a Robotized “da Vinci” Surgical System; a new multicolor angiographer; a highly innovative robotic platform for rapid ex vivo drug testing. In addition, INT-Pascale has strategically invested in technological upgrading the radiation therapy unit, a high-specialty center, providing the structure several sophisticated instruments including a CyberKnife® System for precision radiotherapy.

Education
In 2013 it has concluded the process for the final accreditation of the INT – Pascale as National Provider CME. At the INT – Pascale are active both a Graduate Degree Course in Nursing and a Graduate Degree Course in Biomedical Laboratory Techniques.














